Introduction
In the United States, breast cancer is the most common cancer in women. Men can also develop this pathology, but it is less common, accounting for less than 1% of all cases of breast cancer.
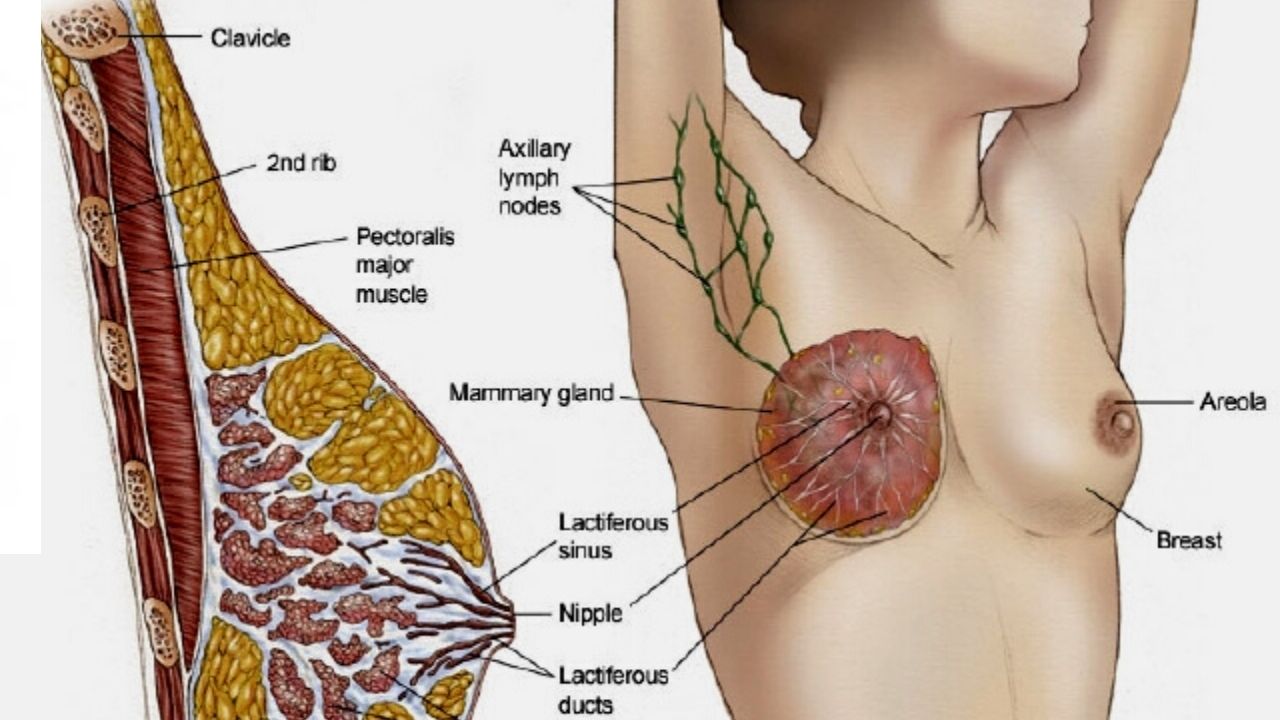
Breast anatomy
The mammary gland is made up of different tissues, from soft (fatty) to very dense. This tissue is made up of lobes, each of which is made up of small tubular structures (lobules). They contain glands that produce milk. Narrow ducts connect the glands, lobes, lobules, carrying milk from the lobes to the nipple. The nipple is located in the center of the field, the dark area (areola).
Blood and lymph nodes pass through the entire mammary gland. Blood feeds cells. The lymphatic system removes metabolic end products from the body. The lymphatic vessels are connected to the lymph nodes – small bean-shaped organs that help fight infections.
About breast cancer
Cancer begins when healthy breast cells change and grow uncontrollably, forming a mass called a tumor. It can be cancerous and benign. Cancer is malignant, meaning it can grow and spread to other parts of the body. The benign neoplasm grows but does not spread to other tissues.
Breast cancer can spread to other parts of the body. Also, malignant cells travel to other parts of the body through the blood and lymphatic vessels. This phenomenon is called metastases.
Most often, oncology spreads to nearby lymph nodes, but it can further spread through the body to the bones, lungs, liver and brain. This is called metastatic cancer or stage IV breast cancer.
If the malignant process returns after primary treatment, it can occur locally, that is, in the mammary gland and / or regional lymph nodes. Regional lymph nodes are lymph nodes located near the breast, such as in the armpit. Also, oncology can reappear in any other organ, this will be called a distant relapse or metastatic relapse.
Types of breast cancer
Breast cancer is invasive and non-invasive. Invasive cancer is a process that spreads to surrounding tissues. Non-invasive cancer does not extend beyond the milk ducts or lobules. In most cases, cancer begins in the ducts or lobes and is called ductal carcinoma or lobular carcinoma:
- Ductal carcinoma is a type of malignant neoplasm that begins in the cells lining the milk ducts and accounts for the majority of cancers.
- Ductal carcinoma in situ (DCIS). Cancer that is found only in the duct.
- Invasive or infiltrative ductal carcinoma. A tumor that has spread beyond the duct.
- Invasive lobular carcinoma. A tumor originating from a lobule.
Less common types of breast cancer include:
- Medullary
- Slime-forming
- Tubular
- Metaplastic
- Tumors of the nipple
- Inflammatory carcinoma of the breast is a fast growing cancer, accounting for 1 to 5% of all breast cancers.
- Paget’s disease is a type of cancer that occurs from the ducts in the nipple. Usually developing in situ, Paget’s disease is, however, also invasive.
Breast cancer subtypes
There are 3 main subtypes of oncology, which are determined by special analyzes of the tumor sample. These tests can help your doctor learn more about your cancer and recommend the most effective treatment plan.
Analyzes can determine the following subspecies of the oncological process:
- The hormone receptor is positive. Breast cancer that expresses the estrogen (ER) and / or progesterone (PR) receptors is called hormone receptor positive. They are proteins found in cells. Tumors containing estrogen receptors are called ER-positive, progesterone-PR-positive. To define cancer as a hormone receptor positive, it must be positive for only one of these receptors. The growth of such a neoplasm may be dependent on the hormones estrogen and / or progesterone. Hormone receptor positive cancer occurs at any age, but is more common in postmenopausal women. About 60-75% of breast cancers have estrogen and / or progesterone receptors. Cancer without these receptors is called hormone receptor negative.
- HER 2 is positive. The growth in approximately 10-20% of all breast cancers depends on a gene called human epidermal growth factor receptor 2 (HER 2). These types of cancer are called HER2-positive and have many copies of the HER 2 gene or high levels of the HER 2 protein. These proteins are also called receptors. HER 2 geneforms the HER2 protein, which is found in malignant cells and is important for their growth. HER2-positive breast cancer grows faster. These tumors can also be hormone receptor positive or hormone receptor negative. Cancers that have little or no HER2 protein and / or few copies of the HER2 gene are called HER2 negative.
- Triple negative. If the tumor does not express ER, PR, or HER2, it is termed triple negative. It accounts for 15 to 20% of invasive breast cancers. This type of oncology is more common in young women, especially in blacks and hispanics. Triple negative cancer is also more common in women with a mutation in the BRCA1 or BRCA genes. Experts recommend that all people with a similar pathology under the age of 60 be tested for BRCA gene mutations.
Risk factors and prevention
A risk factor is anything that increases the likelihood of developing a disease. Although risk factors often influence the development of cancer, most of them do not. Knowing your own risk factors and discussing them with your doctor can help you make more informed lifestyle and health care choices.
Most cancers are sporadic, that is, they occur due to accidental damage to a person’s genes after birth. There is no risk of passing this gene on to children.
Hereditary cases of breast cancer are less common, accounting for 5 to 10% of cancers. It develops with changes in genes, called mutations, and is passed in the family from parents to children.
Many of these mutations occur in tumor suppression genes such as BRCA 1 or BRCA 2. These genes usually prevent cells from spiraling out of control and becoming cancerous. But when mutations occur in cells, they can get out of control.
When analyzing the oncological process of the breast, it is important to remember that the majority of women with such a pathology did not have obvious risk factors and a family history of breast cancer.
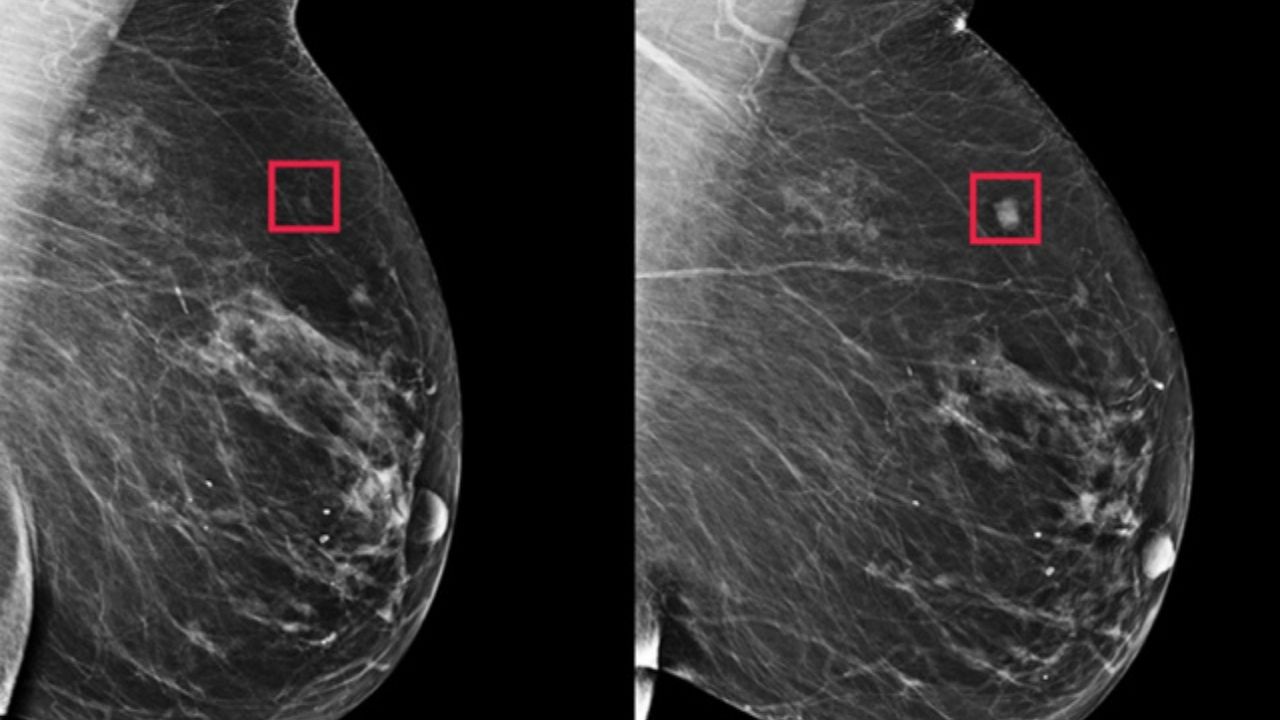
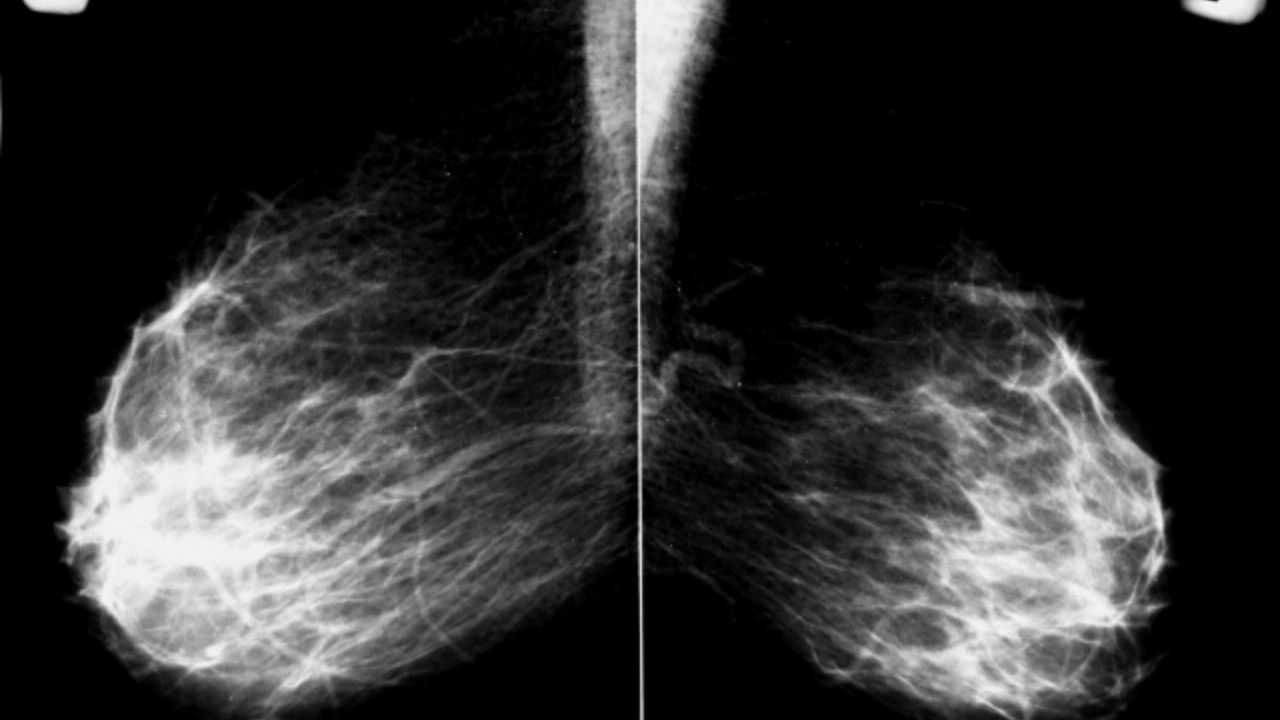
Every woman should talk with her doctor about getting regular breast exams and mammograms. A mammogram is an X-ray of the mammary glands that can detect a tumor that is too small and therefore undetectable by touch.
The following factors may increase a woman’s risk of developing breast cancer:
Age. The risk of developing breast cancer increases with age, with most cases being more common in women over 50.
Anamnesis of life. Women who have had cancer in one breast have a high risk of developing cancer on the other breast.
Family history of breast cancer. Malignant neoplasm of one breast can occur in families in the following situations:
- If one or more women have breast cancer diagnosed at age 45 or earlier.
- One or more women are diagnosed with breast cancer before age 50 and have a family history of other cancers such as prostate cancer, melanoma, and pancreatic cancer.
- Oncology of the breast and / or ovaries has been present in many generations of the same family line, for example, these cancers were diagnosed in the paternal grandmother and aunt.
- The woman is re-diagnosed with breast and ovarian cancer.
- Breast cancer was diagnosed in a male relative.
- Family history of breast, ovarian, prostate and / or pancreatic cancers on the same lineage.
- The presence of relatives belonging to the Ashkenazi ethnic group.
If your family has any of the above situations, it is important to discuss it with your doctor. This could be a sign that a genetic mutation for breast cancer, such as BRCA1 or BRCA2, is present in your family (see the Hereditary Risk / Genetic Susceptibility section below).
It is also important to consider your father’s side when looking at your family history. It is just as important as the mother’s side in determining an individual’s risk of developing breast cancer.
Hereditary risk / genetic predisposition. There are several inherited genes associated with an increased risk of breast cancer and other cancers. The most famous mutations are those in BRCA1 or BRCA2. Mutations in these genes are associated with an increased risk of breast and ovarian cancer and other cancers.
The risk of breast cancer in men, as well as the risk of prostate and other cancers, is also increased if there are mutations in these genes.
Other gene mutations or inherited diseases can also increase your risk of developing breast cancer. They are much less common than BRCA1 or BRCA2 and do not increase risks as much.
- Lynch syndrome associated with genes MLH1, MSH2, MSH6 and PMS2
- Cowden’s syndrome (SC) associated with the PTEN gene
- Li-Fraumeni syndrome associated with the TP53 gene
- Peutz-Jegers syndrome (SPE) associated with the STK11 gene
- ataxia-telangiectasia (AT) associated with the ATM gene
- Hereditary diffuse gastric cancer associated with the CDH1 gene
- PALB2 gene
- CHEK2 gene
There are other genes that increase the risk. Research is also ongoing to find other genes that may affect breast cancer risk.
A genetic blood test can identify known mutations in the BRCA1 and BRCA2 genes and other genes associated with inherited syndromes. Your doctor may recommend a test called a screening test. It aims to look for mutations in several different genes at the same time.
The doctor may also suggest texts, but they are not recommended for everyone; they are prescribed only after the person has received appropriate genetic counseling.
History of ovarian cancer. Mutations in the BRCA1 and BRCA2 genes significantly increase the risk of ovarian and breast cancer. Consequently, women who are diagnosed with hereditary ovarian cancer caused by a BRCA gene mutation also have an increased risk of developing breast cancer.
Women with breast cancer who have not inherited the BRCA1 or BRCA2 mutation usually do not have a higher risk of developing adnexal neoplasms.
Early menstruation and late menopause. Women who start menstruating before the age of 11-12 or who have menopause after age 55 have a slightly higher risk of developing cancer. This is because their breast cells have been exposed to estrogen and progesterone for a longer time.
Estrogen and progesterone are female hormones that control the development of secondary sex characteristics such as breast development and pregnancy. The production of estrogen and progesterone in women decreases with age, and sharply decreases during menopause. Long-term exposure to these hormones increases the risk of developing cancerous tumors.
Time of pregnancy. Women who become pregnant after 35, or women who have never had a full-term pregnancy, have a higher risk of developing breast cancer. Being pregnant can protect a woman from cancer, as it promotes the transition of breast cells to the final stage of maturity.
Hormone replacement therapy after menopause. The use of hormone therapy (based on estrogen and progesterone) after menopause, often called hormone replacement therapy, within the previous 5 years or over several years, increases the risk of cancer. The number of diagnosed breast cancers has dropped significantly as fewer women are taking hormone replacement therapy.
However, women who take only estrogen, without taking progesterone, for 5 years (due to removal of the uterus or for other reasons) have a slightly lower risk of developing breast cancer.
Oral contraceptives, or birth control pills. Some studies show that oral contraceptives do not significantly increase the risk of breast cancer.
Race and Ethnicity. Breast cancer is the most common form of cancer in women. White women are more prone to breast cancer than dark-skinned women, but among women younger than 45, the disease is more common in blacks than in whites.
Cancer deaths are also higher in black women. Causes of differences in mortality rates may include biological characteristics, other health conditions, and socioeconomic factors that affect access to health care.
Ashkenazim, or the Eastern European group of Jews, also have an increased risk of developing breast cancer, as they have a higher risk of BRCA mutations. Breast cancer is less common among Hispanics, Asia-Pacific, American Indian and Alaskan women.
Atypical breast hyperplasia. This diagnosis increases the risk of developing cancer in the future. It is characterized by the presence of abnormal, but not cancerous, cells found on a breast biopsy.
Lobular carcinoma in situ (DCIS). DCIS stands for abnormal cells found in the lobules, or glands, of the breast. It is not considered cancer.
However, DSIS in one breast increases the risk of developing invasive breast cancer in the future. If DCIS is found on a biopsy, it can be removed to check for other changes. Additional treatment may be recommended.
Density of the breast. Dense breast tissue can make it difficult to detect cancer with standard imaging tests such as mammography. Tighter tissue usually means more glands, milk ducts, and supporting tissue than adipose tissue.
Breast tightness is also due to higher estrogen levels and is not a risk factor in and of itself, and usually decreases with age.
Lifestyle. Research continues to show that various aspects of lifestyle can contribute to the development of breast cancer, just like other cancers.
- The weight. Recent studies have shown that postmenopausal women who are overweight or obese have an increased risk of developing breast cancer. These women are also more likely to have cancer recurrence.
- Physical activity. Reduced physical activity is associated with an increased risk of breast cancer and an increased likelihood of relapse after treatment. Regular physical activity can protect against cancer by helping to maintain a healthy weight, lowering hormone levels, or causing changes in metabolism or immune factors.
- Alcohol. Modern research shows that drinking more than 1-2 servings of alcoholic beverages per day, including beer, wine and spirits, increases the risk of cancer, as well as the risk of relapse after treatment.
- Nutrition. There is no reliable research to prove that eating or avoiding certain foods reduces the risk of breast tumors or cancer recurrence after treatment. However, consuming more fruits and vegetables and less animal fat is associated with many health benefits.
Socio-economic factors. Higher income women in all racial and ethnic groups have a higher risk of developing breast cancer than lower income women in the same groups. The reasons for this difference are still unknown.
But they are associated with differences in nutrition, environmental factors, and other risk factors, such as breast density. Women living in poverty are more likely to be diagnosed at an advanced stage and are less likely to survive the disease than women who are wealthier.
This is likely due to several factors, including lifestyle factors and other health characteristics such as obesity and tumor biology. Access to health care and the availability of treatment also play an important role.
Exposure to radiation at an early age. Exposure to ionizing radiation at a young age can increase a woman’s risk of developing breast cancer. For example, therapeutic radiation to the chest area in Hodgkin’s lymphoma increases the risks.
The very small amount of radiation a woman receives during mammography is not associated with an increased risk of cancer.
How do you assess your risk of developing breast cancer?
Several breast cancer risk assessment tools have been developed to help a woman assess her likelihood of developing breast cancer. The best studied is the Gale model, available on the official website of the National Cancer Institute at www.cancer.gov/bcrisktool.
After entering some personal and family information, including race / ethnicity information, the tool will provide you with a 5-year and lifetime risk assessment for developing invasive breast cancer.
It only asks for information on breast cancer from first-degree family members and does not take into account their age at the time of diagnosis, so it is best suited for assessing risk in women without a high risk of hereditary cancer.
Prevention of breast cancer
There are different risk factors for each type of cancer. Although there is no proven way to completely prevent breast cancer, the risk can be reduced.
The following methods can help reduce this risk.
Surgery to reduce the risk of cancer
Women with BRCA1 or BRCA2 genetic mutations that significantly increase their risk of breast cancer may consider prophylactic breast removal. This procedure, called prophylactic mastectomy, reduces the chance of developing a tumor by at least 95%. Women with these mutations should also consider prophylactic removal of the ovaries and fallopian tubes, called prophylactic salpingo-oophorectomy.
This procedure can reduce the risk of ovarian cancer and possibly breast cancer by stopping the production of estrogen by the ovaries.
Drugs that lower the risk of cancer
Women with a higher risk of developing cancer may talk with their doctor about taking medications that can help prevent breast cancer. This approach is called “endocrine prophylaxis” or “chemoprophylaxis”. For breast cancer, it consists of using hormonal drugs to reduce the risk of cancer.
- Tamoxifen (available in generic form) is a drug called a selective estrogen receptor modulator (SERM). It is often used as a treatment for breast cancer in women who have already developed the disease. Tamoxifen blocks the effects of estrogen on tumor growth. Tamoxifen may serve as a way to reduce the risk of cancer, especially ER-positive breast cancer in women over 35. Studies have shown that it may be effective and cause fewer side effects in women with carcinoma in situ or a precancerous breast condition called atypical hyperplasia. It is not recommended for women with a history of blood clots, smokers, and long periods of immobility (unable to move). It is also not recommended for pregnant women, women with suspected pregnancy and lactating. Side effects of tamoxifen may include hot flashes, vaginal discharge, sexual discomfort, mood changes, and an increased risk of uterine cancer, blood clots, and stroke.
- Raloxifene (Evista) is also a SERM. It is often prescribed to prevent osteoporosis (thinning of the bones) in postmenopausal women. Raloxifene may be suitable for women over 35 with an increased risk of breast cancer during menopause. Raloxifene can be used for more than 5 years, it may reduce the risk of thinning bones, as well as the risk of breast cancer. It is not recommended for women who have not reached menopause or have a history of blood clots, smoke, and have been deprived of mobility for a long time. Side effects of raloxifene may include hot flashes, leg cramps, swelling of the hands and feet, weight gain, vaginal dryness, and pain during intercourse.
- Aromatase inhibitors (AIs). AIs reduce the level of estrogen in a woman’s body, stopping its production. To reduce the risk of developing breast cancer, 2 AIs are used: exemestane (aromazine) and canastrozole (arimidex). AIs can also be used as an alternative to tamoxifen in women at high risk of developing cancer during menopause. AI is not recommended for women before menopause and is generally not recommended for women with osteoporosis. Side effects of AIs include hot flashes, fatigue, sleep problems, diarrhea, muscle and joint pain, vaginal dryness, and thinning of the bones.
Lifestyle changes to reduce cancer risk
One way to reduce your risk of breast cancer is through regular physical activity. Studies show that 30-60 minutes a day of moderate to high intensity physical activity reduces the likelihood of developing cancer.
Other ways are to maintain a healthy weight, limit the amount of alcohol consumed, and skip hormone replacement therapy to reduce symptoms during menopause. Breastfeeding can also reduce the risk of breast cancer in women.
Follow-up
Treatment for people diagnosed with cancer does not end once active therapy is completed. Your doctors will continue to check for cancer recurrence, monitor for side effects, and monitor your overall health. This is called follow-up.
It can include regular medical check-ups, medical tests, or both. Doctors are keen to track your progress over the months and years to come. In some cases, patients will be able to visit clinics for cancer survivors. These clinics specialize in meeting the post-treatment needs of people diagnosed with breast cancer.
Relapse control
One of the goals of follow-up is to control relapse, that is, return of the disease. Cancer will recur because small areas of undetected malignant cells may remain in the body. Over time, these cells can grow until they appear on test results or cause signs or symptoms.
Breast cancer can recur in the breast or other areas of the body. Typically, a relapse is detected when a person develops symptoms or abnormal signs during a physical examination.
Symptoms depend on where the cancer has recurred and may include:
- Lump under the arm or along the chest wall.
- Constant pain that gets worse and is not relieved by OTC drugs.
- Pain in bones, back, neck or joints, fractures or swelling, possible signs of bone metastases.
- Headaches, seizures, dizziness, confusion, personality changes, loss of balance, changes in vision, possible signs of brain metastases.
- Chronic cough, shortness of breath or trouble breathing, possible symptoms of lung metastases.
- Abdominal pain, itching or rash on the skin, or jaundiced skin and eyes due to a condition called jaundice, which may be associated with liver metastases.
- Changes in energy, such as feeling unwell or extremely tired.
- Poor appetite and / or weight loss.
- Nausea or vomiting.
It is important that your surgeon explains to you how the sensations in the chest will change after surgery and radiation therapy, if any.
Over time, the breasts will soften and change. If you have questions or concerns about your symptoms, you should discuss them with your doctor.
Control of long-term and delayed side effects
Most people expect side effects during treatment. The unpleasant consequences that persist after treatment are called long-term. Delayed side effects can develop months or even years later. Long-term and delayed effects can include physical and emotional changes.
Talk with your doctor about the risk of these side effects, depending on the type of cancer, your individual treatment plan, and your overall health.
If you are aware that your treatment may have some delayed effects, you may need to have certain physical examinations, scans, or blood tests to detect and correct them.
- Long-term effects of surgery . After a mastectomy or lumpectomy to treat early breast cancer or locally advanced cancer, the breast may be scarred, and its shape and size may change. Or, the area around the surgical site may become hardened. If the lymph node was removed during surgery or changed as a result of treatment, lymphatic edema may occur in the armpit, in the chest wall or in the chest even many years after treatment. This remains a lifelong risk for survivors.
- Long-term effects of radiation therapy. Some patients after radiation therapy experience shortness of breath, dry cough, and / or chest pain 2-3 months after the end of therapy. This is because radiation therapy can cause swelling and hardening or thickening of the lungs called fibrosis. These symptoms may resemble pneumonia, but they do not go away with antibiotics. Symptoms are treated with drugs called steroids. Most patients recover completely after treatment.
- Long-term effects of trastuzumab and / or chemotherapy. Heart problems may develop in patients who have taken trastuzumab or some types of chemotherapy called anthracyclines. Talk to your doctor about the best way to check your heart condition. They are also at risk of other long-term effects such as menopausal symptoms, fatigue, brain chemistry, persistent numbness and tingling in fingers and toes, and weight gain.
- Long-term effects of hormone therapy. Women taking tamoxifen should have an annual pelvic check-up, as these drugs slightly increase the risk of developing uterine cancer. If you notice unusual vaginal bleeding or other new symptoms, tell your doctor about them, as additional tests may be needed to find out the cause. Women taking AIs such as anastrozole, exemestane, or letrozole should have a bone density test during treatment, as these drugs can cause some weakness and bone wasting.
In addition, women recovering from early breast cancer or locally advanced cancer may have other side effects that persist after treatment ends. However, they can be controlled with medication or other means. After recovery, the following long-term effects are possible:
- menopausal symptoms such as hot flashes;
- joint pain;
- fatigue;
- mood swings.
Vaginal dryness and decreased sex drive are also common side effects of treatment after early breast cancer or locally advanced cancer.
Stages of breast cancer
Staging is determining where the cancer is located, whether it has spread, and where, and how it affects other parts of the body.
Doctors use diagnostic tests to determine the stage of cancer; they may need information based on tissue samples obtained during surgery, so the process may continue until all analyzes have been completed.
Staging information helps the physician determine which treatment is optimal and predict the likelihood of recovery. There is a different stage description for each type of cancer.
TNM tumor classification system
The most common way of staging a cancer is with this TNM system. Doctors use the results of diagnostic tests and scans to answer the following questions:
- Tumor (T): How large is the primary tumor? Where is it?
- Lymph Nodes (N): Has the tumor spread to the lymph nodes? If so, where and to what extent?
- Metastasis (M): Has the cancer spread to other parts of the body? If so, where and to what extent?
To determine the stage of the oncological process for each patient, the results are combined.
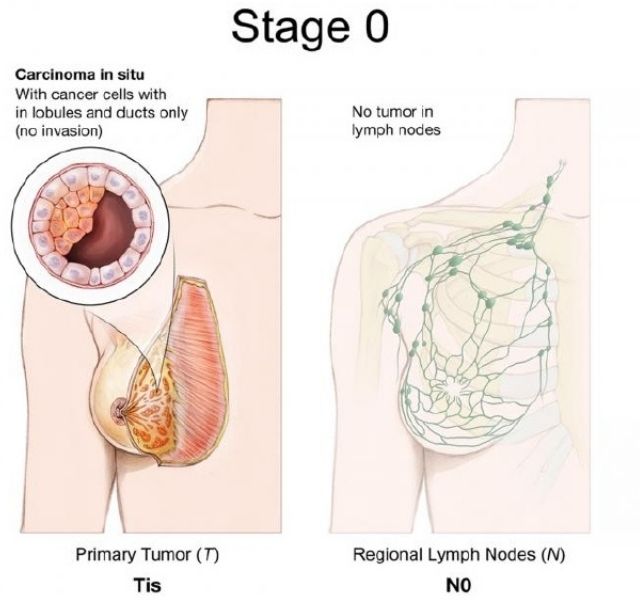
There are 5 stages: stage 0 (zero), which is non-invasive ductal carcinoma in situ (DCIS), and stages I through IV (1 through 4), which are used to evaluate invasive breast cancer.
The stage provides a general way of describing oncology so that clinicians can work together to plan optimal treatment.
The stages can be clinical and pathological. The clinical classification is based on pretreatment examinations, including physical examinations, mammograms, ultrasounds, and MRIs.
Pathological classification is based on the results of surgery to remove breast tissue and lymph nodes. Results are usually available a few days after surgery. In general, pathological data provide most of the information for determining patient prognosis.
Below is detailed information about each element of the TNM System for Breast Oncology:
Tumor (T)
In TNM, the letter T plus a letter or number (0 to 4) is used to describe the size and location of the tumor. The size of the lesion is measured in centimeters (cm). A centimeter is about the width of a standard pen or pencil.
The stages are also divided into smaller subgroups, which help to further characterize the tumor. Information about the specific stage of the neoplasm is given below.
TX: primary tumor cannot be assessed;
T0 (T plus zero): There is no evidence of a primary breast tumor.
Tis: Carcinoma in situ. The cancer is restricted to the ducts in the breast tissue and has not spread to the surrounding breast tissue. There are 2 types of in situ breast carcinoma:
- Tis (PCIS): PCIS is a non-invasive neoplasm, but if not removed, it can later develop into invasive breast cancer. PCIS means that cancer cells are found in the ducts of the mammary glands, but have not spread outside the tissues in which they originated.
- Tis: Paget’s disease (oncology of the nipple of the breast) is a rare, early-stage, non-invasive cancer that occurs only in the skin cells of the nipple. Sometimes Paget’s disease occurs against the background of another, invasive breast disease. In the presence of another, invasive breast cancer, the disease is classified according to the stage of the tumor.
T1: A breast mass is 20 millimeters (mm) or less in its largest dimension. This stage is divided into 4 substages, depending on the size of the tumor:
- T1mi – 1 mm or less mm;
- T1a – more than 1 mm but less than 5 mm;
- T1b – more than 5 mm but less than 10 mm;
- T1c – more than 10 mm but less than 20 mm.
T2: More than 20 mm but less than 50 mm.
T3: Over 50 mm
T4: The tumor belongs to one of the following groups:
- T4a – The tumor has spread to the chest wall.
- T4b – The tumor has grown into the skin.
- T4c – The cancer has spread to the chest wall and skin.
- T4d – Inflammatory Breast Cancer.
Lymph nodes (N)
The N in the TNM system stands for lymph nodes. Regional lymph nodes include:
- Armpit lymph nodes (axillary lymph nodes)
- Lymph nodes above and below the collarbone;
- The lymph nodes under the breastbone are the internal breast lymph nodes.
Lymph nodes in other parts of the body are called distant. If a doctor evaluates lymph nodes before surgery based on tests and / or physical examination, the letter c is placed before the letter N, from the word “clinical” – clinical. If the doctor evaluates the lymph nodes after surgery, this assessment will be more accurate, and the letter p is placed before the letter N – from the word “pathologic” (pathological). The information below refers to pathologically defined stages.
NX: Lymph nodes are not evaluated.
N0: One of the following conditions is met:
- There are no signs of lymph node cancer.
- The lymph nodes have cancerous areas less than 0.2 mm in size.
N1: Cancer has spread to 1-3 axillary lymph nodes and / or internal mammary lymph nodes. If the cancer in the lymph node is more than 0.2 mm but less than 2 mm, it is micrometastatic cancer (N1mi).
N2: Cancer has spread to 4-9 axillary lymph nodes. Or, the cancer has spread to the internal breast lymph nodes, but not to the axillary
N3: Cancer has spread to 10 or more lymph nodes. Or it has spread to the lymph nodes under the collarbone or sternum. Cancer that has spread to the supraclavicular lymph nodes is also stage N3.
Knowing how many lymph nodes are affected and where they are will help doctors plan treatment. The histologists will be able to find out how many lymph nodes contain cancer after they are surgically removed.
As a rule, the supraclavicular or internal mammary lymph nodes are not removed during the operation. If they are affected, other therapy is used first, such as radiation, hormonal therapy, or chemotherapy.
Metastases (M)
The TNM letter M stands for the spread of cancer to other parts of the body, that is, distant metastases. In this case, it is no longer considered early stage cancer or locally advanced cancer.
MX: Remote metastases cannot be assessed.
M0: The disease has not metastasized.
M0 (i +): No clinical or radiological evidence of distant metastases. Microscopic signs of tumor cells are found in blood, bone marrow or other lymph nodes no larger than 0.2 mm.
M1: There are signs of metastasis to another part of the body, that is, breast cancer cells are growing in other organs.
Cancer stages
Doctors determine the stage of cancer by combining T, N, and M classifications, as well as tumor grade and ER / PR and HER2 test results. A simpler approach to explaining the stage of breast cancer is to use the T, N and M classifications. This approach is used below to describe the different stages.
Most patients want to know the exact stage of the cancer. This is possible after all tests have been carried out, usually 5-7 days after the operation. When systemic treatment or a general treatment called neoadjuvant therapy is given prior to surgery, the staging is initially determined clinically.
Early on, doctors may classify cancer as stage I through IIA, and locally advanced cancer as IIB-III.
Stage 0: Describes a disease that is found only in the ducts of breast tissue and does not spread to the surrounding breast tissue. It is also called non-invasive cancer (Tis, N0, M0).
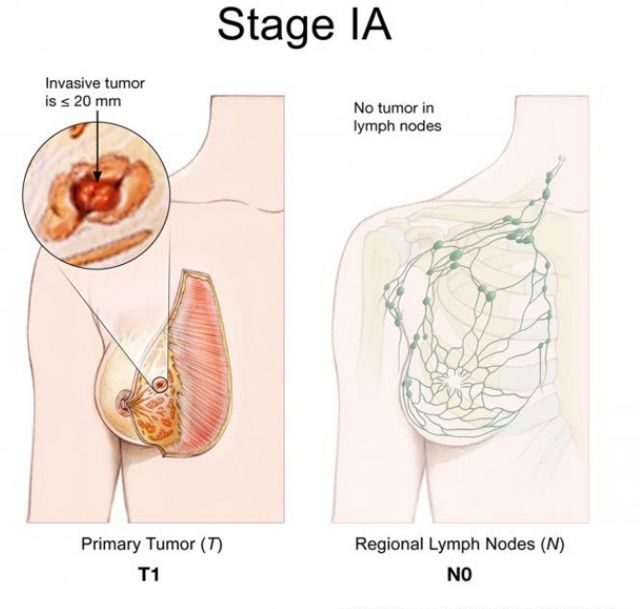
Stage IA: The tumor is small, invasive and has not spread to the lymph nodes (T1, N0, M0).
Stage IB: Cancer has spread to the lymph nodes, is more than 0.2 mm but less than 2 mm. Either there is no evidence of a breast tumor, or the breast tumor is 20 mm or less (T0 or T1, N1, M0).
Stage IIA: One of the following conditions:
- There are no signs of a tumor in the breast, but the cancer has spread to 1-3 axillary lymph nodes. The cancer has not spread to distant parts of the body. (T0, N1, M0).
- The tumor is 20 mm or less and has spread to the axillary lymph nodes (T1, N1, M0).
- The neoplasm is more than 20 mm, but not more than 50 mm and does not extend to the axillary lymph nodes (T2, N0, M0).
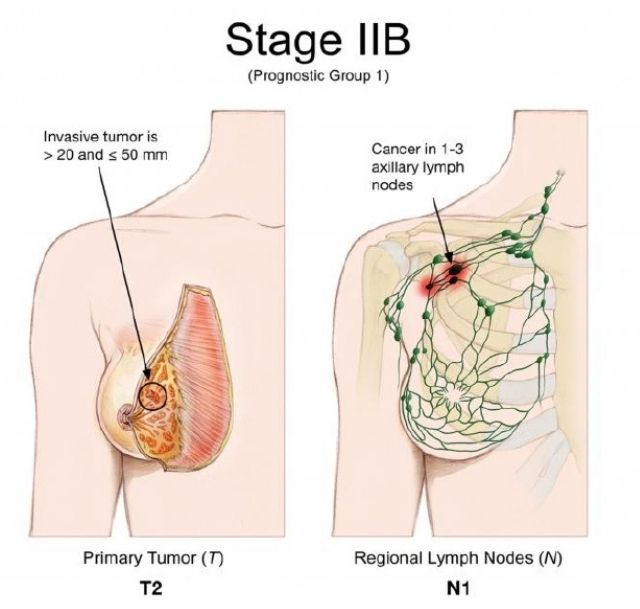
Stage IIB: Any of the following conditions:
- The tumor is more than 20 mm, but not more than 50 mm and spreads to 1-3 axillary lymph nodes (T2, N1, M0).
- The neoplasm is more than 50 mm, but does not spread to the axillary lymph nodes (T3, N0, M0).
Stage IIIA: Cancer of any size has spread to 4-9 axillary lymph nodes or to the inner chest. It has not spread to other parts of the body (T0, T1, T2 or T3, N2, M0). Stage IIIA can also refer to a tumor larger than 50 mm that has spread to 1-3 axillary lymph nodes (T3, N1, M0).
Stage IIIB: The mass has spread to the chest wall or caused swelling or ulceration of the breast, or is diagnosed as inflammatory breast cancer. It can spread to the axillary or internal breast lymph nodes. Does not apply to other parts of the body (T4; N0, N1 or N2; M0).
Stage IIIC: A tumor of any size that has spread to 10 or more axillary lymph nodes, internal breast lymph nodes, and / or lymph nodes under the collarbone. Has not spread to other parts of the body (any T, N3, M0).
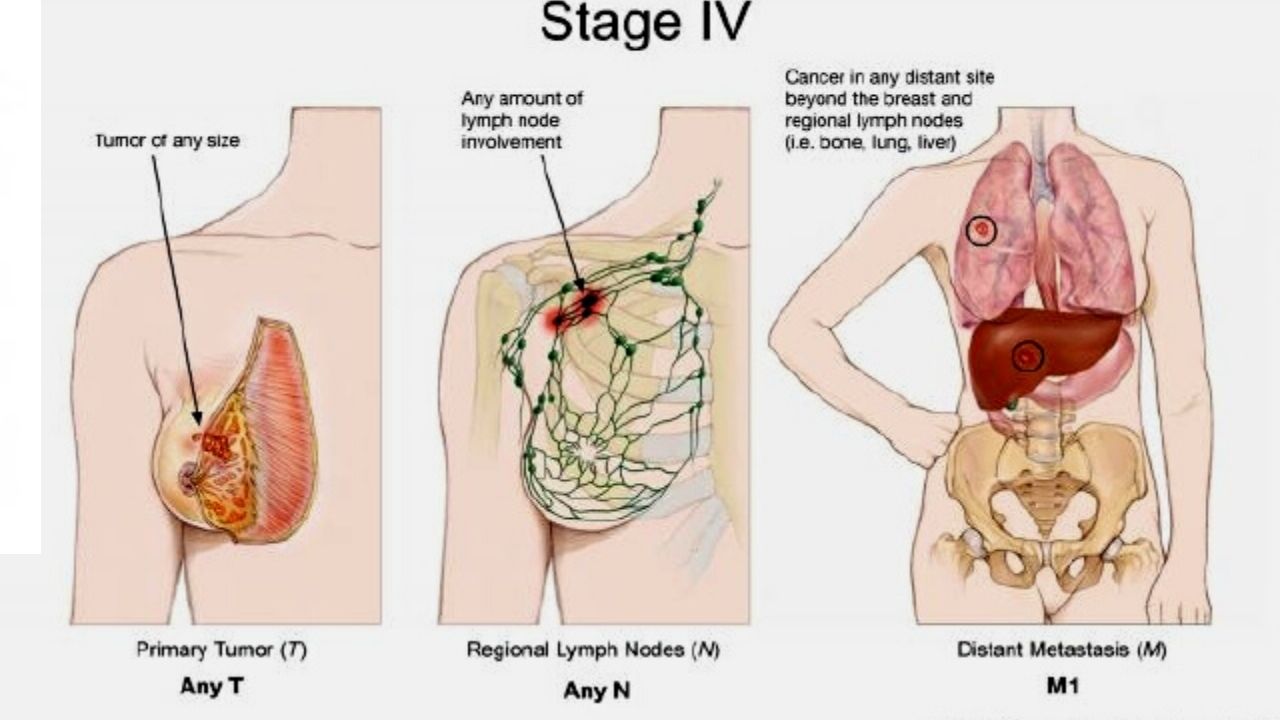
Stage IV (metastatic): The tumor can be of any size and has spread to other organs such as bones, lungs, brain, liver, distant lymph nodes, or chest wall (any T, any N, M1). Metastatic cancer, detected at the first diagnosis of cancer, occurs in about 6% of cases.
It can be called metastatic breast cancer de novo. Most often, oncology with metastases is found after a previous diagnosis of early cancer.
Relapse: Cancer relapse is the return of the oncological process after treatment. It can be local, regional and / or remote. In case of relapse, you will need to pass all tests again to determine the degree of relapse.
Symptoms of breast cancer
Most women do not see any signs or symptoms of breast cancer at the time of diagnosis.
The following are signs and symptoms that should be discussed with your doctor. In many cases, other, non-cancerous conditions may be the cause of the symptom.
- A mass that feels like a hard lump or lump in the breast or armpit. It is important to feel the same area in the other breast to make sure it is not part of healthy tissue in that area.
- Changes in the size or shape of the breast
- Sudden bloody discharge from the nipple or discharge from only one breast
- Physical changes, such as nipple inversion or nipple pain
- Irritation or change in skin condition, such as wrinkled skin, dents, bumps, or wrinkles
- Hot, reddened, swollen mammary gland, with or without rashes, with pits like orange peel, also called lemon peel symptom
- Chest pain, in particular, persistent. Pain is usually not a symptom of breast cancer, but should be reported to your doctor.
If you are concerned about any changes in your body, see your doctor. Your doctor will ask, among other things, how long and how often you have been experiencing the symptom (s). This conversation will help determine the cause of the problem, which is called the diagnosis.
Once cancer is diagnosed, symptom relief remains an important part of cancer care and cancer treatment. This can be called palliative or supportive care. It often begins shortly after diagnosis and continues throughout treatment.
Be sure to talk to your doctors about the symptoms you are experiencing, including any new symptoms or symptom changes.
Treatment methods
This section describes the treatments that are the standard of care for early breast cancer and locally advanced cancer. “Standard of Care” refers to the best known treatment.
When deciding on a treatment plan, you are encouraged to consider clinical trials as an option. This study is testing a new approach to treatment. Doctors want to know if new treatments are safe, effective, and possibly more acceptable than standard treatments.
Clinical trials can test a new drug, a new combination of standard treatments, or new doses of standard drugs or other treatments. Clinical research is an option for treatment and cancer care at all stages of cancer.
Treatment overview
When treating cancer, different doctors specializing in different areas of cancer treatment – surgeons, radiologists, oncologists – work together to develop an overall treatment plan for a patient that integrates different treatments.
This is called an interdisciplinary team. Cancer care teams include doctors, nurses, cancer nurses, social workers, pharmacists, consultants, nutritionists, and other professionals.
Oncologists – gerontologists are involved in the treatment of patients over 65 years of age. The composition of the team may change over time if your treatment plan needs to change.
A treatment plan is a summary of your cancer and its treatment plan. It is designed to provide basic medical history information to all the doctors who will guide you throughout your life.
The biology and behavior of oncology influences the treatment plan. Some tumors are smaller but grow quickly, while others are larger but grow slowly. Treatment options and recommendations are highly individualized and depend on several factors, including:
- From tumor subtype, including hormone receptor (ER, PR) status and HER status
- From the stage of the tumor.
- From genomic markers such as Oncotype DX ™ and MammaPrint ™ (as required).
- From the patient’s age, general health, menopausal status and preferences.
- For known mutations in inherited breast cancer genes such as BRCA1 or BRCA
Breast cancer doctors tailor the treatment for each patient and each type of cancer; however, there are some general guidelines for therapy.
For both PCIS and early-stage invasive breast cancer, doctors usually recommend surgery to remove the tumor. To ensure complete removal of the tumor, surgeons also remove a small area of healthy tissue around the tumor, called an edge.
The goal of the surgery is to remove all visible cancer; despite this, microscopic cells in the mammary gland or elsewhere may be missed. In some situations, this means another operation will be required to remove the remaining cancer cells.
For larger or faster-growing masses, doctors may recommend systemic treatment with chemotherapy or hormone therapy before surgery, this is called neoadjuvant therapy. Additional therapy before surgery can have several benefits:
- Women who may need a mastectomy may have breast-conserving surgery (lumpectomy) if the swelling shrinks before surgery.
- The operation may be easier because the swelling will be smaller.
- Your doctor will be able to find out if certain cancer treatments work well.
- You can also try new treatments in clinical trials.
After surgery, the next step in treating early oncology is to reduce the risk of recurrence and get rid of any remaining cancer cells. They are undetectable but are thought to be responsible for cancer recurrence as they can grow over time.
Treatment after surgery is called adjuvant therapy. Adjuvant therapy can include radiation therapy, chemotherapy, tartet and / or hormonal therapy.
The need for adjuvant therapy depends on the likelihood of retaining cancer cells in the breast or in the body in general, and on the likelihood that certain treatments will be able to cope with cancer treatments.
Adjuvant therapy reduces the risk of recurrence, but does not eliminate the risk entirely.

In addition to staging, there are activities aimed at determining the prognosis and helping in making decisions about adjuvant therapy. These activities include tumor tissue tests that can predict the risk of recurrence (such as Oncotype Dx ™ or MammaPrint ™). These tests can help your doctor better understand your cancer risk and the ability to reduce those risks with chemotherapy.
If it is impossible to remove the cancer with the help of surgery, such oncology is called inoperable. In this case, doctors recommend treating it in other ways. Chemotherapy, targeted therapy, radiation therapy, and / or hormone therapy may be given to shrink the tumor.
For recurrent cancer, treatment options depend on how the process was previously treated and on the above characteristics of the disease, in particular ER, PR and HER2.
The following is a description of the standard treatment options for early breast tumors and locally advanced cancers. Your plan may also include treating symptoms and side effects, which are also an important part of cancer treatment.
Surgery
Surgical treatment is the removal of the tumor and surrounding healthy tissue through surgery. The operation is also needed to examine the nearby axillary lymph nodes. An oncologist surgeon is a doctor who specializes in the treatment of cancer with surgical methods.
Typically, the smaller the tumor, the more options the patient has for surgery. There are the following options for the operation:
- Lumpectomy. This is the removal of the tumor and a small free edge of healthy tissue around the tumor. Most of the breast remains. For invasive cancer, postoperative radiation therapy is usually recommended for the remaining breast tissue. For PCIS, postoperative radiotherapy may be appropriate, depending on the patient and the tumor. Lampiectomy is also called breast conserving surgery, partial mastectomy, quadranttectomy, or segmental mastectomy.
- Mastectomy. This is the surgical removal of the entire breast. There are several types of mastectomy. Talk to your doctor about preserving the skin – preserving the skin – or the nipple – preserving the nipple-areola complex.
Removal and analysis of lymph nodes
In some cancers, malignant cells are found in the axillary lymph nodes. It is important to understand if there is cancer in the lymph nodes in the breast area. This information is used to determine the type of treatment and prognosis.
- Sentinel lymph node biopsy . In a sentinel lymph node biopsy, the surgeon locates and removes a small number of lymph nodes in the armpit that are receiving drainage from the mammary glands. This procedure avoids the removal of multiple lymph nodes with axillary resection in patients whose sentinel lymph nodes are predominantly cancer-free. Removing fewer lymph nodes helps reduce the risk of some side effects. These side effects include swelling of the hand, called lymphostasis ,the risk of numbness, as well as problems with motor function of the arm and limitation of shoulder mobility, which remain for a long time and significantly reduce the person’s quality of life. The histologist then examines these lymph nodes for the presence of cancer cells. To locate the sentinel lymph node, the surgeon will usually inject dye and / or radioactive tracer behind or around the nipple. The injection, which may cause some discomfort, takes about 15 seconds. The dye or indicator enters the lymph nodes, first of all entering the sentinel node. If a dye is used, the surgeon can find the color change he is looking for. If a radioactive tracer is used, it emits radiation that helps the surgeon find.If the sentinel node does not contain cancer, then, according to research, it is likely that the remaining lymph nodes will also not contain it. This means that there is no need to remove other lymph nodes. If only 1 or 2 sentinel nodes contain cancer, and you are planning lumpectomy and radiation therapy for the entire breast, axillary lymph node removal may not be necessary.
- Removal of axillary lymph nodes. Many lymph nodes are removed under the armpit. They are then examined by a histologist for the presence of cancer cells. In fact, the number of lymph nodes removed may vary. Axillary removal may not be necessary for all women with early breast cancer with few cancer cells in the sentinel lymph nodes. If cancer is found in the sentinel lymph node, the need for additional surgery to remove more lymph nodes depends on the specific situation.
Most women with invasive breast cancer will have a sentinel lymph node biopsy or axillary lymph node removal.
However, sometimes these procedures are not necessary for patients over 65 years of age. It depends on the size of the nodes, the stage of the tumor, and the general health of the person.
A sentinel lymph node biopsy is not done if there are obvious signs of cancer in the lymph nodes before surgery. In this situation, complete removal of the axillary lymph nodes is preferable.
Typically, lymph nodes are not evaluated in patients with PKIS and non-invasive cancer because the risk of cancer spread is very low. However, the surgeon may consider a sentinel lymph node biopsy in patients with PKIS who decide to have a mastectomy or need it.
Reconstructive (plastic) surgery
Women after a mastectomy may consider breast reconstruction. This is a breast reconstruction surgery using tissue taken from another part of the body or synthetic implants.
Reconstruction is usually performed by a plastic surgeon. Sometimes it is possible to perform reconstruction at the same time as the mastectomy, defining it as immediate. Sometimes the reconstruction is performed later, calling it deferred.
For patients with lumpectomy, plastic surgery can be performed at the same time to improve the appearance and shape of the breasts. It is called oncoplastic.
Many mammologist surgeons can perform it without the help of plastic surgeons. Surgery on a healthy breast is sometimes also suggested to make both breasts look the same.
The following methods are commonly used to form a new breast.
Implants. A breast implant is a form filled with saline or silicone gel to reshape the breast. Outside, the saline-filled implant consists of silicone and is filled with sterile saline, that is, salt water.
Silicone gel implants are filled with silicone instead of saline. They were previously thought to cause connective tissue damage, but no clear evidence of this has been found. Before permanent implants are placed, a woman may be temporarily placed with a tissue expander to create a pocket of the correct size.
Talk to your doctor about the benefits and risks of using silicone and saline implants. The lifespan of the implant varies from woman to woman. Some women never have to change them. Other important factors to consider when choosing an implant include:
- Saline implants sometimes “wrinkle” at the top or dislocate over time, but many women find it easy to move them.
- Saline implants feel different from silicone implants. They are often harder than silicone.
Sometimes breast implants lead to problems. Some women have problems with their shape or appearance. Implants can rupture or break, cause pain, and lead to scarring or infections around the implant.
They are rarely associated with other cancers. These problems occur frequently, however, you should discuss the risks with your doctor.
Closure with tissue flap. This technology uses muscles and tissues from other parts of the body to restore the shape of the breasts. Tissue graft surgery is performed from a “leg flap,” that is, tissue from the back or abdomen is grafted onto the chest without cutting off the blood vessels.
A “free flap” means that the blood vessels are cut off and the surgeon needs to transplant tissue onto new blood vessels in the chest. There are the following tissue graft procedures:
- Transverse rectus abdominis muscle (TRAM) flap. This technique, performed with a pedunculated flap or free flap, uses muscle and tissue from the lower abdominal wall.
- Latissimus dorsi flap. With this method of graft graft, muscle and tissue are taken from the upper back.
- Flap – deep inferior epigastric artery perforator (DIEP) flap. For the DIEP free flap, tissue is taken from the abdomen and the surgeon sutures the blood vessels to the chest wall.
- Free flap from the buttocks. The flap is taken from the buttocks to repair the muscles and tissues of the breast, and the surgeon also sutures the blood vessels.
Blood vessels are involved in the flap closure, so this strategy is usually not recommended for women with diabetes, connective tissue or vascular diseases, for women who smoke, as they have a higher risk of problems during and after surgery.
DIEP and free buttock grafts take longer and also take longer to recover. However, the appearance of the breasts can be a priority, especially when radiation therapy is part of the treatment plan.
External breast simulators (prostheses)
An external breast prosthesis, or an artificial breast, is a suitable choice for women who are planning breast surgery in the future or not at all.
They are made of silicone or soft material and are inserted into the bra after a mastectomy. Breast prostheses can be ideal and provide a natural look for every woman.
Summary of surgical procedures
Summing up, the following surgical treatment options can be distinguished:
- Breast cancer removal: Lumpectomy, or partial mastectomy, usually followed by radiation therapy in cases of invasive cancer. With PCIS, radiotherapy may or may not be used. A mastectomy with or without immediate reconstruction may also be recommended.
- Lymph node assessment: Sentinel node biopsy and / or axillary node removal.
More aggressive surgeries, such as mastectomy, are not always more suitable and may cause more complications. The combination of lumpectomy and radiation therapy results in a slightly higher risk of cancer recurrence in the same breast or in the surrounding area.
However, the long-term survival rate for women choosing lumpectomy is exactly the same as for women after mastectomy. Even with a mastectomy, not all breast tissue may be removed and there is a chance of recurrence.
Women with a very high risk of developing cancer in the other breast may consider bilateral mastectomy, that is, removal of both breasts. This applies to women with BRCA1 or BRCA2 gene mutations and women with cancers of both glands.
For women who are not at very high risk of developing new cancers in the future, removing healthy breasts with bilateral mastectomy does not prevent cancer recurrence and does not increase the chances of survival.
Although the risk of developing new cancer in this gland will be reduced, surgery to remove the other breast does not reduce the risk of the original cancer recurring. In addition, more extensive surgery can be associated with a greater risk of problems.
Radiation therapy
Radiation therapy is the use of high-energy X-rays or other particles to destroy cancer cells. A doctor who specializes in performing radiation therapy to treat cancer is called a radiation oncologist. There are several types of radiation therapy:
- External beam therapy. this is the most common option, and the radiation emanates from the apparatus outside the body.
- Intraoperative radiation therapy – when radiation therapy is performed with a probe in the operating room.
- Brachytherapy. With this type of radiation therapy, radioactive sources are placed inside the tumor.
Despite encouraging research results, intraoperative radiation therapy and brachytherapy have not been widely adopted. If possible, they can be used in patients with a small tumor that has not spread to the lymph nodes.
A radiotherapy regimen usually consists of a specified number of treatments performed over a specified period of time.
Radiation therapy often helps reduce the risk of breast cancer recurrence. In fact, with modern surgery and radiotherapy, the recurrence rate of oncology is currently less than 5% within 10 years after treatment, and the survival rate is the same as with lumpectomy or mastectomy.
If cancer is present in the lymph nodes in the armpit, radiation therapy may also be given on the same side of the neck or armpits near the breast or chest wall.
Radiotherapy can be given after or before surgery:
- Adjuvant therapy is given after surgery. It is most often prescribed after lumpectomy and sometimes chemotherapy. Patients after mastectomy may not require radiation therapy, depending on the characteristics of the tumor. Radiotherapy may be recommended after a mastectomy for a large tumor, cancer in the lymph nodes, cancer cells outside the lymph node capsule, or cancer that has invaded the skin or chest wall, or for other reasons.
- Neoadjuvant therapy is radiation therapy given before surgery to shrink a large tumor to make it easier to remove. This approach is not very common and is considered only when the tumor cannot be removed by surgery.
Radiation therapy can cause side effects, including fatigue, breast swelling, redness and / or discoloration of the skin, and pain or burning in the area of the skin where the radiation is directed, sometimes blistering or scaling.
Your doctor may recommend topical skin preparations to treat some of the side effects.
In the past, there has been a slight increase in the long-term risk of heart disease in women treated with left-sided breast cancer with older equipment and radiation therapy.
Modern methods now eliminate most of the heart disease from the list of side effects of radiation therapy.
Radiation therapy scheme
Typically, radiation therapy is applied daily for a specified number of weeks.
- After a lumpectomy. post -lumpectomy radiation therapy is external beam radiation therapy given daily, Monday through Friday, for 3-4 weeks if the cancer is not in the lymph nodes. If the cancer is in the lymph nodes, radiotherapy is given for 5-6 weeks. Typically, it starts with radiation therapy for the entire breast and then moves on to more specific treatment of the tumor site before the end of the course of treatment.
This highly targeted portion of treatment, called increased radiation dose, is the standard of care for women with invasive breast cancer to reduce the risk of breast recurrence. - After a mastectomy. Women who require radiation therapy after a mastectomy are given sessions 5 days a week for 5-6 weeks. Radiotherapy is given before or after reconstructive surgery.
Studies have also been conducted on shorter circuits; they are used in some centers, including in the form of accelerated partial irradiation of the breast (see below) for 5 days.
Such short regimens are not suitable for patients requiring post-mastectomy radiation therapy or lymph node irradiation. In addition, some women with very large breasts require longer radiation therapy regimens.
Partial irradiation of the breast. Partial breast irradiation (POMI) is radiation therapy directed directly to the area of the tumor, rather than the entire breast. Directing the radiation directly to the tumor usually shortens the time of radiation therapy.
However, PRC is only suitable for some patients. Although the early results seemed very promising, PPRs are still being studied. PLC is performed with standard external beam radiation therapy directed to the area from which the tumor was removed, rather than the entire breast.
PLCS can also be performed with brachytherapy using plastic catheters or metal probes temporarily placed inside the breast. Breast brachytherapy does not last long (1 dose to 1 week). It can consist of 1 dose, administered directly in the operating room directly after the removal of the tumor.
- Intensity modulated radiation therapy (IMRT). It is a more advanced method of breast radiation therapy. The intensity of the radiation directed to the breast can be varied to better target the tumor, spreading the radiation more evenly through the breast. Immunization with IMRT reduces the dose of radiation and can reduce the potential damage to nearby organs, such as the heart and lungs, and the risks of some side effects. IMRT may also help reduce the long-term effects on breast tissue, such as induration, swelling, and discoloration, which were very common with older radiation therapy. IMR is not recommended for everyone. Discuss this with your radiologist for more information.
- Proton therapy. Standard radiation therapy for breast cancer uses X-rays to kill cancer cells, also called photon therapy. Proton therapy is a form of external beam radiation therapy that uses protons rather than x-rays. Due to their high energy, protons can destroy cancer cells. Protons have different physical properties, due to which radiation therapy can be more targeted than photon therapy, and the radiation dose can potentially be reduced. This therapy also reduces the amount of radiation passing near the heart. Researchers are studying the benefits of proton therapy over photon therapy in a national clinical trial.
Features of adjuvant radiation therapy for older patients and / or patients with small tumors
Recent studies have looked at the possibility of avoiding radiotherapy for women over the age of 70 with an early ER-positive tumor or for women with a small tumor.
Overall, these studies showed that radiation therapy reduced the risk of breast cancer recurrence in the same breast compared to no radiation therapy. However, radiation therapy does not make women live longer.
Drug therapy
Systemic therapy is the use of medications to kill cancer cells. These drugs are injected through the bloodstream to reach cancer cells throughout the body. Systemic therapy is usually prescribed by an oncologist, a doctor who specializes in treating cancer with drugs.
Common medical treatments include inserting an intravenous (IV) catheter into a vein with a needle or swallowing (oral) tablets or capsules.
For breast cancer, the following types of systemic therapy are used:
- Chemotherapy;
- Hormone therapy;
- Targeted therapy;
- Immunotherapy.
Each of these types is described in more detail below. A person can receive 1 type of systemic therapy or a combination of several types at the same time. They can also be part of a treatment plan that includes surgery and / or radiation therapy.
Chemotherapy
Chemotherapy is the use of drugs that destroy cancer cells, preventing them from growing and producing new cells. It can be given before surgery to shrink large tumors, facilitate surgery, and reduce the risk of recurrence, and is called neoadjuvant chemotherapy.
It can also be given after surgery to reduce the risk of recurrence, then this is called adjuvant chemotherapy.
A chemotherapy regimen, or schedule, usually consists of a combination of drugs given over several cycles of treatments over a period of time.
Chemotherapy is carried out according to different regimens, depending on which one has shown itself best in clinical trials. It can be done once a week, once every 2 weeks, once every 3 weeks, or even once every 4 weeks.
There are many types of chemotherapy used to treat breast cancer. The most common drugs are:
- Capecitabine (Xeloda)
- Carboplatin (available in generic form)
- Cisplatin (available in generic form)
- Cyclophosphamide (available in generic form)
- Docetaxel (Taxotere)
- Doxorubicin (available in generic form)
- Pegylated Liposomal Doxorubicin (Doxil)
- Epirubicin (Ellens)
- Eribulin (halaven)
- Fluorouracil (5-FU)
- Gemcitabine (Gemzar)
- Ixabepilone (Ikzempra)
- Methotrexate (rheumatrex, trexall)
- Paclitaxel (Taxol)
- Protein-linked paclitaxel (abraxane)
- Vinorelbin (Navelbin)
The patient can receive 1 type of systemic therapy or a combination of several types at the same time.
Studies have shown that combinations of several drugs for adjuvant therapy are sometimes more effective than a single drug. The following drugs or drug combinations can be used as adjuvant therapy for early breast cancer or locally advanced breast cancer:
- AC (doxorubicin and cyclophosphamide)
- EC (epirubicin, cyclophosphamide)
- AC or EC (epirubicin and cyclophosphamide), then T (doxorubicin and cyclophosphamide, then paclitaxel or docetaxel, or vice versa)
- CAF (cyclophosphamide, doxorubicin and 5-FU)
- CEF (cyclophosphamide, epirubicin and 5-FU)
- CMF (cyclophosphamide, methotrexate and 5-FU)
- TAC (docetaxel, doxorubicin and cyclophosphamide)
- TC (docetaxel and cyclophosphamide)
Therapies targeting the HER2 receptor can be used with chemotherapy for HER2-positive breast cancer. An example is the trastuzumab antibody. A combination regimen for HER2-positive early breast cancer may include:
- AC-TH (doxorubicin, cyclophosphamide, paclitaxel or docetaxel, trastuzumab)
- AC-THP (doxorubicin, cyclophosphamide, paclitaxel or docetaxel, trastuzumab, pertuzumab)
- TCHP (paclitaxel or docetaxel, carboplatin, trastuzumab, pertuzumab)
- TCH (paclitaxel or docetaxel, carboplatin, trastuzumab)
- TH (paclitaxel, trastuzumab)
The side effects of chemotherapy depend on the individual tolerance, the drug (s) used, the regimen and dose.
These can include fatigue, risk of infection, nausea and vomiting, hair loss, loss of appetite, diarrhea, constipation, numbness and tingling in the fingers, early menopause, weight gain, and brain chemistry.
These side effects can often be successfully prevented or reduced with maintenance medications, and they usually go away after treatment ends.
Rarely, long-term effects can occur, such as heart problems, permanent nerve damage, or secondary cancers such as leukemia and lymphoma. Many patients feel well during chemotherapy and actively care for their families, work and sports, although each patient has different feelings.
Hormone therapy
Hormone therapy, also called endocrine therapy, is effective for most tumors that are estrogen or progesterone receptor positive (also called ER positive or positive or PR positive).
This type of tumor uses hormones to stimulate growth. Blocking hormones, whether used alone or after chemotherapy, will help prevent cancer recurrence and death from cancer.
Hormone therapy may be given before surgery to shrink tumors, facilitate surgery, and reduce the risk of recurrence, and is called neoadjuvant hormone therapy.
In addition, it can be given after surgery to reduce the risk of recurrence, then this is called adjuvant hormone therapy.
Types of hormone therapy
- Tamoxifen. Tamoxifen is a drug that blocks the binding of estrogen to breast cancer cells. It is effective in reducing the risk of recurrence, the risk of developing cancer in the other breast, and the risk of long-term recurrence. Tamoxifen works well in postmenopausal and pre-menopausal women and is taken as an oral tablet. It is important to discuss any other medications or supplements you are taking with your doctor, as some drugs may interact with tamoxifen. Common side effects of the drug include hot flashes as well as vaginal dryness, discharge, or bleeding. Very rarely, cancer of the uterine lining, cataracts and blood clots develop. However, tamoxifen can improve bone health and lower cholesterol levels.
- Aromatase inhibitors (AIs).AIs reduce estrogen production in all tissues except the ovaries in postmenopausal women by blocking the aromatase enzyme. This enzyme converts weak male hormones called androgens into estrogens when the ovaries stop producing estrogen during menopause. These drugs include anastrozole (arimidex), exemestane (aromazine), and letrozole (femara). All AIs are presented in pill form, taken by mouth on a daily basis. AIs can only be used by postmenopausal women or those taking medications to stop ovarian estrogen production. Treatment with AI as primary hormone therapy or after treatment with tamoxifen may be more effective than tamoxifen alone in reducing the risk of recurrence in postmenopausal women. Side effects of AI include muscle and joint pain, hot flashes, vaginal dryness, and an increased risk of osteoporosis and fractures of the limbs and high cholesterol levels. Women who have not yet reached menopause and who are not receiving injections to stop ovarian function should not take AIs as they do not block the effects of ovarian estrogen. Often, doctors monitor blood estrogen levels in women who have recently stopped menstruating or who stop menstruating after chemotherapy to make sure the ovaries are no longer producing estrogen. and who do not receive injections to stop ovarian function should not take AIs as they do not block the effects of ovarian estrogen. Often, doctors monitor blood estrogen levels in women who have recently stopped menstruating or who stop menstruating after chemotherapy to make sure the ovaries are no longer producing estrogen. And who do not receive injections to stop ovarian function should not take AIs as they do not block the effects of ovarian estrogen. Often, doctors monitor blood estrogen levels in women who have recently stopped menstruating or who stop menstruating after chemotherapy to make sure the ovaries are no longer producing estrogen.
- Suppression of ovarian function, or ablation. Ovarian suppression is the use of medications to stop the ovaries from making estrogen. Ovarian ablation is surgery to remove the ovaries. These options can be used in addition to another type of hormone therapy in women who have not gone through menopause.
- To suppress ovarian function, gonadotropin or luteinizing hormone-releasing hormone (GnRH or LHRH) drugs are used to stop the ovaries from producing estrogen, which causes temporary menopause. These drugs include goserelin (zoladex) and leuprolide (eligard, lupron). They are given by injection every 1 to 3 months and stop the production of estrogen by the ovaries. The effects of GnRH drugs subside after discontinuation of therapy.
- Ovarian ablation uses surgery to remove the ovaries to stop the production of estrogen. As an irreversible measure, this surgery is a good option for women who are not planning to give birth anymore.
Hormone therapy for women after menopause
For women who have gone through menopause, several options for hormone therapy may be prescribed:
- Tamoxifen for 5-10 years.
- IA within 5-10 years.
- Tamoxifen for 5 years, followed by AI for 5 years. Thus, the total duration of hormone therapy will be 10 years.
- Tamoxifen for 2-3 years, followed by AI for 2-8 years, only 5 to 10 years of hormone therapy.
In general, women with stage 1 cancer should expect 5 years of hormone therapy. Women with stage II or III should expect hormone therapy for up to 10 years.
Hormone therapy for women before menopause
As noted above, premenopausal women should take more than AI as they won’t work. In the premenopausal period, there are the following options for adjuvant therapy:
- Tamoxifen for 5 years. Treatment will then depend on whether they have had menopause in those 5 years.
- If a woman does not have menopause after 5 years of treatment, she can continue taking tamoxifen for another 5 years, this will be only 10 years.
- If a woman goes through menopause within the first 5 years of treatment, she can continue to take tamoxifen or switch to AI for the next 5 years. This will only add up to 10 years of hormone therapy. Only women who are clearly menopausal should consider taking an AI.
- Suppression of ovarian function for 5 years with additional hormonal therapy such as tamoxifen or AI may be recommended in the following situations, depending on the woman’s age and the risk of recurrence:
- Women who are diagnosed with breast cancer at a very early age.
- Women who are at very high risk of relapse.
- Women with stage II or III cancer, when, in addition, chemotherapy is recommended.
- Women with stage I or II cancer who are at very high risk of recurrence and who, in addition, may also consider chemotherapy.
- Women who cannot take tamoxifen for other health-related reasons, such as a history of blood clots.
- Ovarian suppression is not recommended as an adjunct to other hormone therapy in the following cases:
- Women who are not at very high risk of cancer recurrence.
- Women with stage I cancer when chemotherapy is not recommended.
Targeted therapy
Targeted therapy is treatment that targets specific genes, proteins, or the tissue environment of a cancer that promotes cancer growth and survival. This type of treatment blocks the growth and spread of cancer cells while limiting damage to healthy cells.
Not all tumors have the same target. To find the most effective treatment, your doctor may run tests to identify genes, proteins, and other factors in your tumor. In addition, research is helping to learn more about specific molecular targets and new therapies that target them.
The first approved targeted drugs for the treatment of breast cancer were hormonal drugs. HER2-targeted therapy was then approved for the treatment of HER2-positive breast cancer.
HER2 targeted therapy
- Trastuzumab (available in generic form). This drug is approved for the treatment of HER2-positive non-metastatic breast cancer. Currently, patients with stage I-III breast cancer must receive a trastuzumab-based regimen, which often includes a combination of this drug with chemotherapy, followed by a total of 1 year of adjuvant trastuzumab. Patients receiving trastuzumab have a small (2 to 5%) risk of heart problems. This risk is increased if the patient has other risk factors for cardiovascular disease or is taking chemotherapy, which also increases the risk of heart disease. These heart problems can go away and can be treated with medication.
- Pertuzumab (perjeta). Approved for the treatment of stage II-III breast cancer in combination with trastuzumab and chemotherapy.
- Neratinib (nerlinks). Approved for the treatment of early stage HER2-positive breast cancer. It is taken for a year, starting 1 year after taking trastuzumab.
- Ado-trastumumab emtansine or T – DM 1 (cadcyl). Approved for early-stage cancer patients treated with trastuzumab and chemotherapy with paclitaxel or docetaxel followed by surgery. T-DM1 is a combination of trastuzumab with a very small amount of strong chemotherapy. This allows the drug to deliver drugs to cancer cells while reducing the effects of chemotherapy on healthy cells, which usually means fewer side effects. T-DM1 is given intravenously every 3 weeks.
Preparations that improve the quality of bone tissue
Bone-improving drugs block bone breakdown and help strengthen bones. They can be used to prevent cancer from recurring to the bone . Certain types of drugs are also used in low doses to prevent and treat osteoporosis (thinning bones)
There are 2 types of drugs that block bone breakdown:
- Bisphosphonates. They block cells that destroy bones called osteoclasts.
- Denosumab (xdzheva). An osteoclast-targeted therapy called a RANK ligand inhibitor.
For people with advanced breast cancer, taking bisphosphonates after breast cancer treatment may help prevent recurrence.
Other targeted therapies for breast cancer
There are other options for targeted therapies for breast cancer, depending on several factors. Many of the following drugs are used for advanced or metastatic breast cancer.
- Alpelisib (picray). Alpelisib is a targeted therapy option along with hormone therapy with fulvestrant for people with HER2-negative metastatic breast cancer with a PIK3CA gene mutation and deterioration during or after hormone therapy.
- Drugs targeting the CDK4 / 6 protein in breast cancer cells, which can stimulate the growth of cancer cells. These drugs include Abemacyclib (Verzenio), Palbociclib (Ibransa), and Ribocyclib (Kisqali). They are approved for women with ER-positive, HER2-negative progressive, or metastatic breast cancer and can be combined with certain types of hormone therapy.
- Lapatinib (Tyerbe) for women with HER2-positive progressive or metastatic cancer, when other drugs are no longer able to control cancer growth. It can be combined with Capecitabine chemotherapy, Lerozole hormone therapy, or HER2-targeted trastuzumab therapy.
- Larotrectinib (Vitrakvi) for the treatment of a confluent NTRK breast tumor that is a metastatic condition or is not resolved by surgery and has been exacerbated by other treatments.
- Olaparib (Linpraza). This oral drug can be used in patients with metastatic HER2 negative breast cancer and a BRCA 1 or BRCA 2 gene mutation who have previously received chemotherapy. This type of drug is called a PARP inhibitor, and it destroys cancer cells, preventing them from repairing damage.
- Talazoparib (Talzenna) for women with locally advanced or metastatic HER2-negative breast cancer and a BRCA 1 or BRCA 2 gene mutation.
Immunotherapy
Immunotherapy, also called biological therapy, is designed to increase the body’s natural defenses against cancer. It uses substances produced by the body or produced in a laboratory to improve, direct or restore the function of the immune system.
- Atezolizumab (Tecentrik). The combination of atzolizumab with protein-coupled paclitaxel has been used successfully for locally advanced triple-negative breast cancer. It is also the only approved breast cancer drug that tests positive for PD-L1.
- Petrolizumab (Keytruda). It is an FDA-approved immunotherapy for the treatment of metastatic cancer or cancer that cannot be treated with surgery. These tumors also have molecular changes called microsatellite high frequency instability (MSI-H) or deficiency in DNA replication error repair (DMMR).
Different types of immunotherapy can cause different side effects. Common side effects include skin reactions, flu-like symptoms, diarrhea, and weight changes.
Features of systemic therapy in elderly patients
Age should not be the only determinant of treatment. Systemic treatments, such as chemotherapy, often work just as effectively in older patients as in younger patients. However, older patients are more likely to experience side effects that affect their quality of life.
It is important for patients to discuss recommended systemic therapy options with doctors, including their benefits and risks. In addition, one should ask about possible side effects and how to eliminate them.
Physical, emotional and social consequences of cancer
Cancer and cancer treatments cause physical symptoms and side effects, as well as emotional, social, and financial consequences. Managing all of these effects is called palliative or supportive care.
Palliative care aims to improve your well-being during treatment by managing symptoms and supporting the non-medical needs of patients and their families.
Anyone, regardless of age, type and stage of cancer, can receive such help. It is most effective when started immediately after cancer is diagnosed. People receiving palliative care along with cancer treatment often experience less severe symptoms, have a better quality of life, and report that they are more satisfied with the treatment.
Palliative care is very diverse and often includes medication, dietary changes, relaxation techniques, emotional and spiritual support, and other therapies. Supportive therapy in some cases can be considered chemotherapy, surgery, radiotherapy.
Research has shown that some integrative or alternative therapies can help manage symptoms and side effects. Integrative medicine is the combined use of cancer medication with alternative treatments such as psychophysical practice, natural foods and / or lifestyle changes. This includes:
- Music therapy, meditation, stress management and yoga to reduce anxiety and stress.
- Meditation, relaxation, yoga, massage and music therapy for depression and other mood problems.
- Meditation and yoga to improve the overall quality of life.
- Acupressure and acupuncture to combat nausea and vomiting during chemotherapy.
Patients may worry about whether treatment might affect their sexual health and ability to have children in the future, and if so, how. We recommend that you discuss these topics with your doctors before starting treatment.
Breast cancer recurruence
If the cancer comes back after treating the disease early, it is said to be recurrent.
May occur in the following parts of the body:
- In the same place as originally. This is called local recurrence ..
- In the chest wall or in the lymph nodes in the armpit or chest. This is called regional recurrence.
- Elsewhere, including distant organs such as bones, lungs, liver, and brain. This is called distant, or metastatic, relapse.
When breast cancer recurs, a new series of tests should be done to learn as much as possible about it. In addition, an additional biopsy may be required to confirm cancer recurrence and to obtain information about the characteristics of the cancer.
After testing, you should discuss possible treatment with your doctor. The treatment plan often includes the therapies described above (surgery, radiation therapy, radiation therapy, chemotherapy, targeted therapy, and hormone therapy).
They can be used in different combinations or with different intensities. The choice of treatment for breast cancer depends on the following factors:
- Previous treatment for primary cancer.
- Time since initial diagnosis.
- The site of recurrence.
- Tumor characteristics such as ER, PR, and HER status
Treatment options for recurrent local or regional breast cancer
Local or regional recurrence is often corrected or curable. Treatment options are described below:
- For women with local recurrence in the mammary gland after initial treatment with lumpectomy and adjuvant radiation therapy, mastectomy is recommended. Typically, with this treatment, the cancer is completely cured.
- For women with local or regional breast recurrence after primary mastectomy, chemical removal of the recurrent tumor is recommended, followed by radiotherapy of the breast and lymph nodes. However, if radiation therapy has already been done for primary cancer, this option may not work. Typically, radiation therapy is not given more than once with a full dose in the same area. Systemic surgery is sometimes done before surgery to shrink the tumor and make it easier to remove.
- Other treatments, including radiation therapy, chemotherapy, hormonal therapy, and targeted therapy, are used to reduce the likelihood of a distant relapse. They are used depending on the type of tumor and the type of treatment previously received.
Whichever treatment plan you choose, palliative care goes a long way in reducing symptoms and side effects. Your doctor may suggest that you participate in clinical research investigating new treatments for this recurrent cancer.
* Refrences;
Expert opinion
In the United States, breast cancer is the most common cancer in women. Men can also develop this pathology, but it is less common, accounting for less than 1% of all cases of breast cancer.